After a median follow-up of 53.7 months, the median overall survival for patients was 57.6 months for the docetaxel arm and 47.2 months for the ADT alone arm. For patients with low volume disease, as defined by no visceral disease and 3 bony metastases or less, no overall survival (OS) benefit was seen with chemotherapy. Abiraterone was studied in mHSPC based on LATITUDE and STAMPEDE which also showed OS benefit over placebo for patients with mHSPC.1,2 One difference between abiraterone and docetaxel is that recent data presented at ESMO 2018 show that abiraterone has OS benefit in all subgroups, including both high and low volume disease.
Apalutamide is a potent androgen receptor antagonist (AR), similar to enzalutamide, and prevents AR nuclear translocation and transcription of AR gene targets. Apalutamide has already shown benefit over placebo in the non-metastatic castration-resistant setting with SPARTAN, a double-blind, placebo-controlled, phase 3 trial involving men with nonmetastatic castration-resistant prostate cancer (nmCRPC) and a prostate-specific antigen doubling time of 10 months or less. In this presentation, Kim Chi, MD, presented data from the TITAN study involving use of apalutamide for patients with mHSPC.
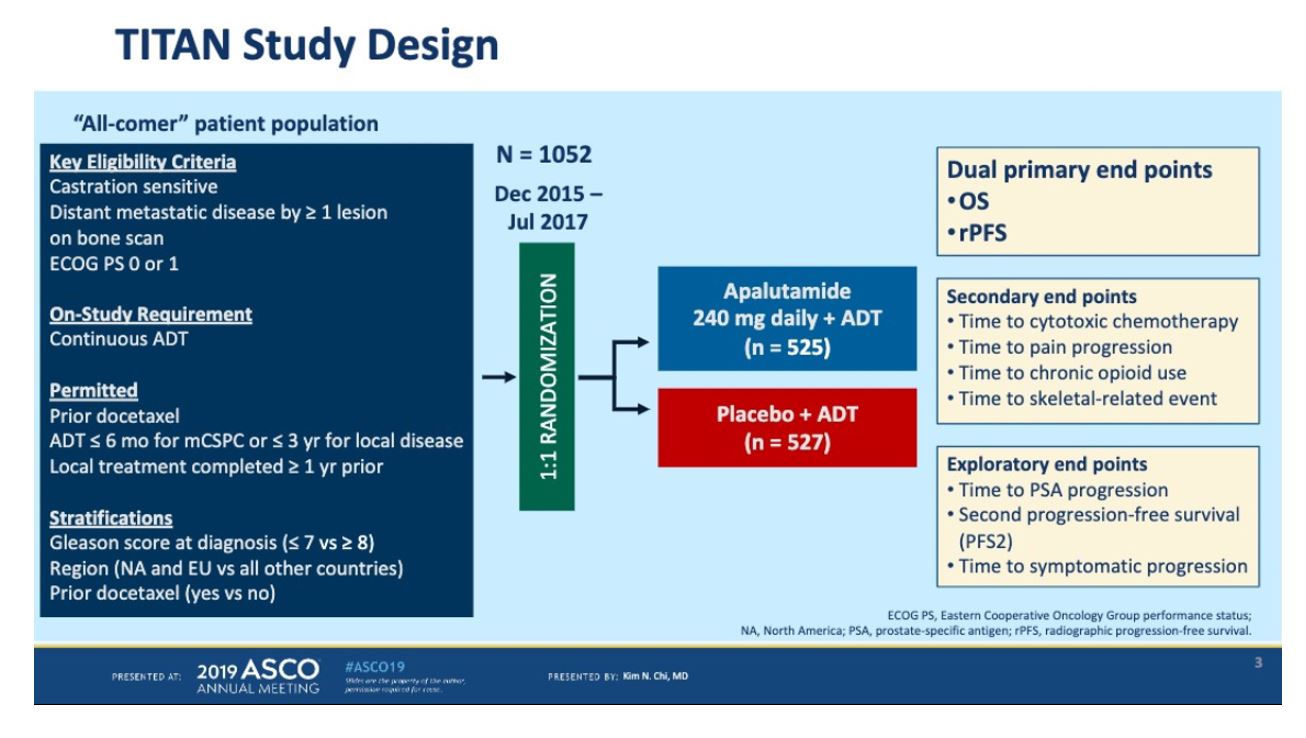
This abstract provides data on 525 patients who were randomly assigned to apalutamide or placebo in a 1:1 ratio. Prior treatment with docetaxel was allowed. The majority of patients had high volume disease (63%) and 11% had prior docetaxel. The primary endpoint was radiographic progression-free survival and overall survival.

Patients were well balanced between the two arms. 10% of patients had received prior docetaxel for mHSPC.

Follow-up times are shown above. 66% of patients remain on treatment on Apalitamide + ADT.
In terms of median radiographic progress free survival, this was 22.1 months in the placebo group and not yet reached in the apalutamide group. Apalutamide improved overall survival (HR = 0.67; 95% CI, 0.51-0.89; p = 0.0053), with a 33% reduction in risk of death and median OS has not yet been reached. Based on these results, the independent data monitoring committee recommended allowing crossover of placebo to apalutamide. 8% of patients taking apalutamide discontinued due to adverse events.

At two years, there was a 20% difference in the rate of radiographic progression free survival (rPFS), with 68% on apalutamide arm free of progression.

Apalutamide reduces the risk of death by 33%, with 82% of patients alive at 2 years in the apalutamide arm compared with 74% in the placebo arm. The overall survival benefit was also consistent through all subgroups.

Secondary endpoints and exploratory endpoints also favored apalutamide.

75% of patients remain free from prostate specific antigen (PSA) progression at 2 years with apalutamide.

In terms of treatment-emergent adverse events, this was mostly similar. Discontinuation of apalutamide was largely due to rash.

Rash occurred in 37% of patients and was mostly grade 1 and 2.

Presented by: Kim N. Chi, MD, FRCPC, British Columbia Cancer Agency, Vancouver, British Columbia, Canada
Written by: Jason Zhu, MD, Fellow, Division of Hematology and Oncology, Duke University, Twitter: @TheRealJasonZhu, at the 2019 ASCO Annual Meeting #ASCO19, May 31-June 4, 2019, Chicago, IL USA
Clinical Trial Information: NCT02489318
References:
- Fizazi K, Tran N, Fein L, et al. Abiraterone plus prednisone in metastatic, castration-sensitive prostate cancer. New England Journal of Medicine 2017;377:352-60.
- James ND, de Bono JS, Spears MR, et al. Abiraterone for Prostate Cancer Not Previously Treated with Hormone Therapy. New England Journal of Medicine 2017;377:338-51.


