(UroToday.com) On the first day of the American Society for Clinical Oncology (ASCO) Genitourinary Cancer Symposium 2022, Poster Session A focussed on the care of patients with prostate cancer. Dr. Parsons presented a poster describing the results of the IPASS study examining the role of PROSTVAC immunotherapy in patients eligible for active surveillance. Active surveillance has become the standard treatment option for patients with low-risk localized prostate cancer. However, a significant proportion of men who begin active surveillance will have disease progression. The use of early immunotherapy could potentially prevent disease progression for these men.
IPASS was a randomized Phase 2 clinical trial to evaluate the clinical effects of PROSTVAC, a vaccinia/fowlpox viral vector-based immunotherapy that contains PSA and three T-cell costimulatory molecules. The authors enrolled 154 patients with clinically localized, low- or favorable intermediate-risk prostate cancer active surveillance. After enrollment, men were randomized (2:1) to receive 7 doses of subcutaneous PROSTVAC or placebo (empty fowlpox vector) over 140 days. Post-intervention prostate biopsy was performed 7-14 days after the last dose. Participants were followed for 6 months post-treatment. The primary outcome was change from baseline to post-vaccination in CD4 and CD8 T cell infiltration in biopsy tumor tissue while secondary outcomes included changes in prostate biopsy Gleason grade (Grade Group) and serum PSA.
The authors found no differences in CD4 and CD8 densities (count of cells/mm2) in post-treatment biopsy tumor tissue between groups (p = 0.63 and p = 0.75, respectively).
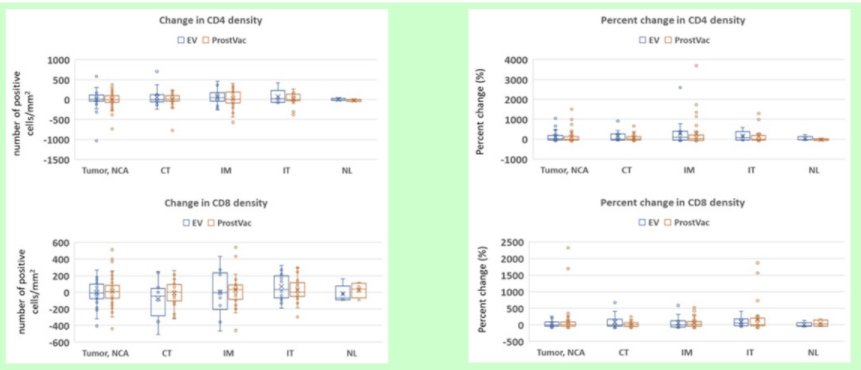
Compared to placebo, patients who received PROSTVAC were less likely to demonstrate upgrading at follow-up biopsy, but this difference did not attain significance (22% vs. 40%, p= 0.08). There was no difference in the change of PSA from baseline to 6 months post-treatment between arms (p= 0.30).
Thus, the authors conclude that, while this first-of-kind trial of immunotherapy for localized prostate cancer demonstrated that PROSTVAC was well tolerated, it did not elicit significant prostate tissue T-cell responses compared to placebo.Presented by: J Kellogg Parsons, MD, MHS, Janssen Research and Development, LLC, San Diego, CA


