(UroToday.com) The 2024 ASTRO annual meeting included a session on novel prognostic tools in prostate cancer, featuring a presentation by Dr. Philip Sutera discussing validation of a digital pathology-based multimodal artificial intelligence (MMAI) model in oligometastatic castration-sensitive prostate cancer, including patients from the STOMP1 and ORIOLE2 phase II randomized trials.
A digital pathology-based MMAI prognostic biomarker (ArteraAI Prostate Test) has been developed and validated within localized prostate cancer and is now an NCCN category 2A recommendation with IB Simon Level of Evidence.3-4 The MMAI biomarker is generated from H&E image features combined with clinical variables including PSA, T stage, and age: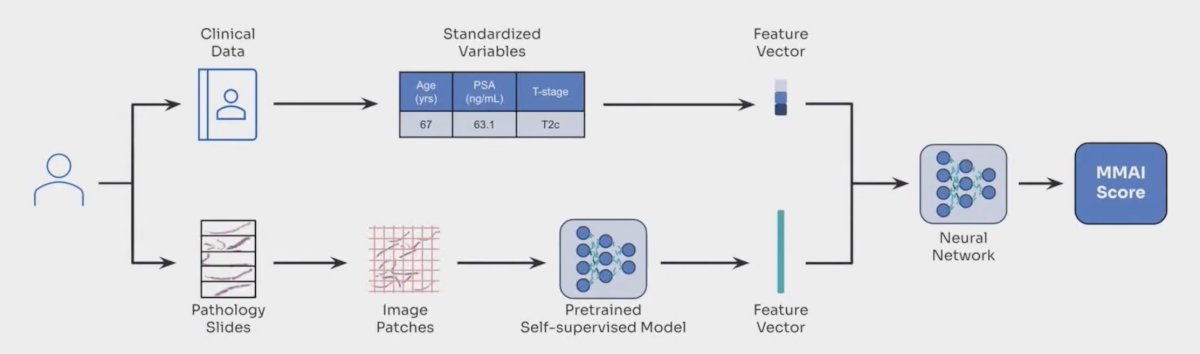
Notably, the MMAI biomarker demonstrated improved prognostication over the NCCN grouping for distant metastasis, prostate cancer-specific mortality, and overall survival. In a recent meta-analysis,5 Spratt et al. showed that among 1,088 men with high-risk disease, those with the MMAI high biomarker were significantly more likely to die of prostate cancer: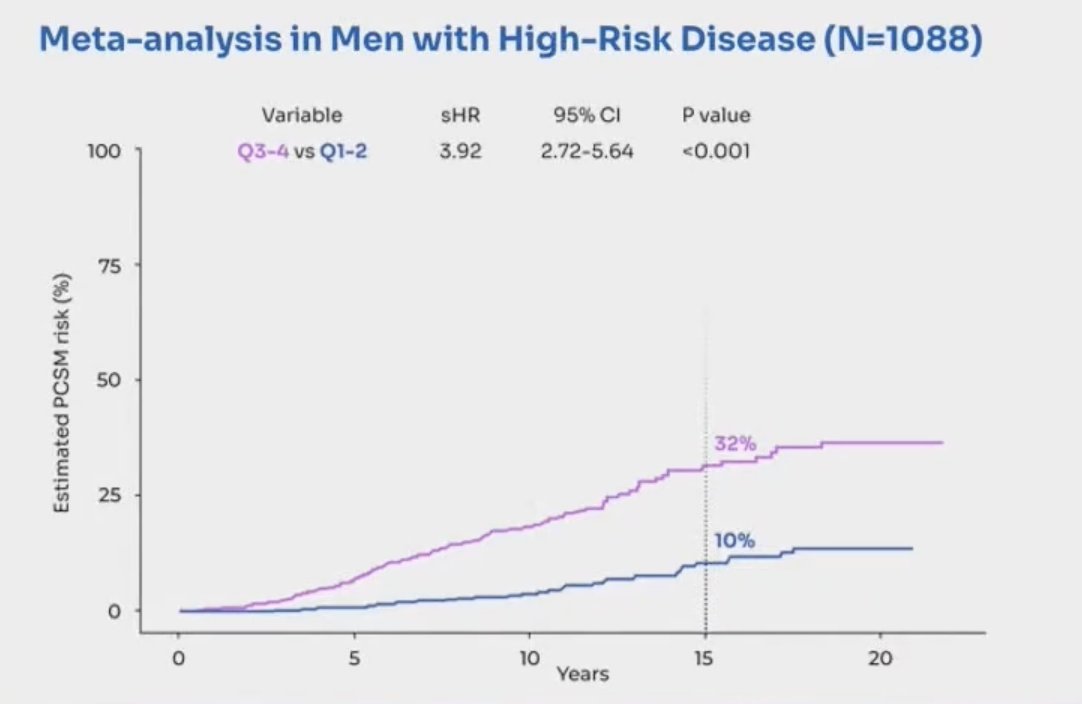
Oligometastatic castration-sensitive prostate cancer is a state of limited metastatic disease. Several randomized trials have demonstrated improvements in progression-free survival in patients with oligometastatic castration-sensitive prostate cancer treated with metastasis-directed therapy. However, clinical outcomes remain heterogeneous, and response to metastasis-directed therapy is variable, raising the need for prognostic and predictive biomarkers. At ASTRO 2024, Dr. Sutera and colleagues evaluated the ArteraAI Prostate Test biomarker in oligometastatic castration-sensitive prostate cancer.
Dr. Sutera and the investigators performed an international multi-institutional retrospective review of 222 men with oligometastatic castration-sensitive prostate cancer (defined as <= 5 metastases) who were evaluated with MMAI scoring. The primary objective was to compare overall survival between patients with high- and low-MMAI scores (stratified by median score). Overall survival was defined as the time from diagnosis of oligometastatic castration-sensitive prostate cancer to death of any cause, calculated with the Kaplan-Meier method and compared using the log-rank test and Cox regression.
The secondary objective was to evaluate MMAI score as predictive for metastasis-directed therapy treatment effect in a subset of patients enrolled in the STOMP and ORIOLE randomized clinical trials. Given too few overall survival events for this subset, they evaluated MMAI for metastasis-free survival, defined as time of randomization to development of a new radiographically detected metastasis or death of any cause, and analyzed using Cox regression. An interaction test was performed between treatment arm and MMAI score.
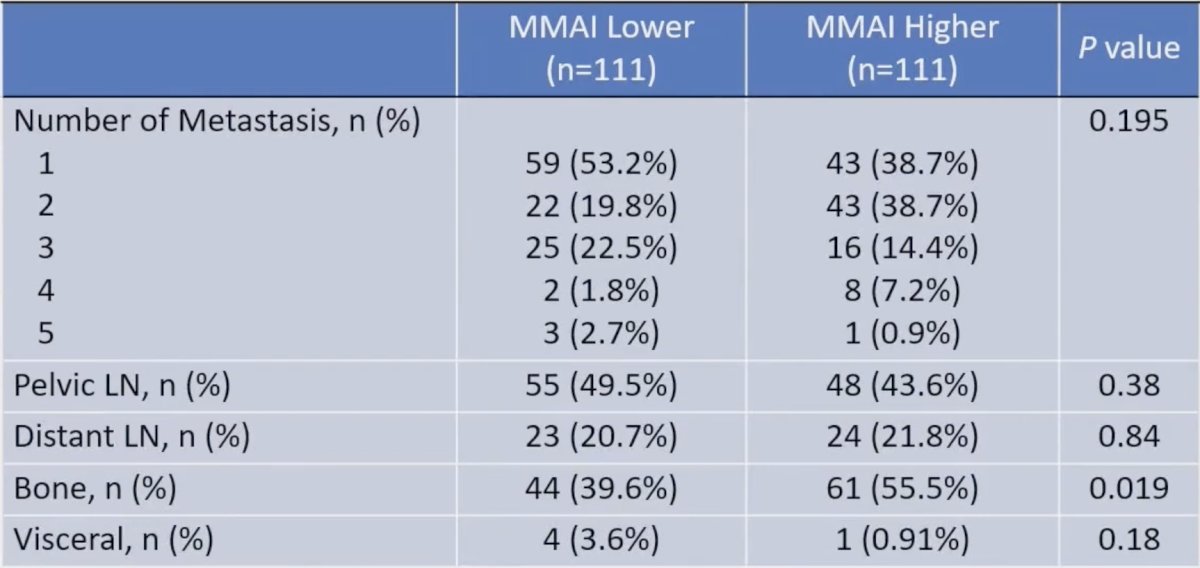
The median follow-up of the surviving patients was 38.0 months. Patients with high MMAI (> 0.527) were found to have higher PSA at diagnosis (9.61 versus 5.95, p < 0.001), higher Gleason score (p < 0.001), more likely to have de novo metastatic disease (28.8% versus 8.1%, p < 0.001), and more likely to have bone metastases (55.5% versus 39.6%, p = 0.019):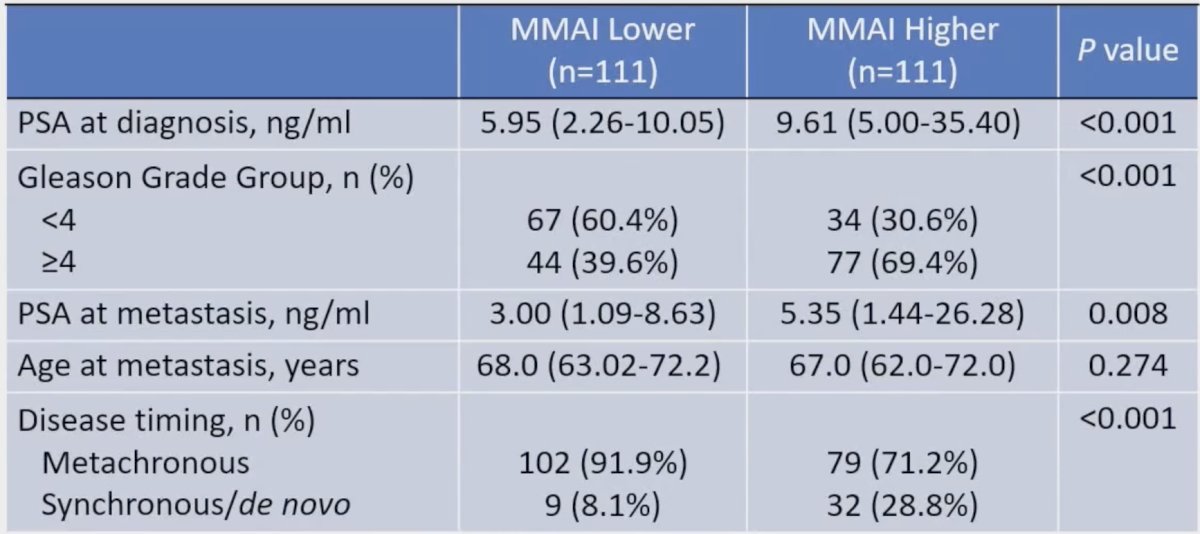
Patients with a high MMAI had a significantly worse overall survival (HR 7.88, 95% CI 1.62-36.49) with a median overall survival of 108.4 months versus not reached: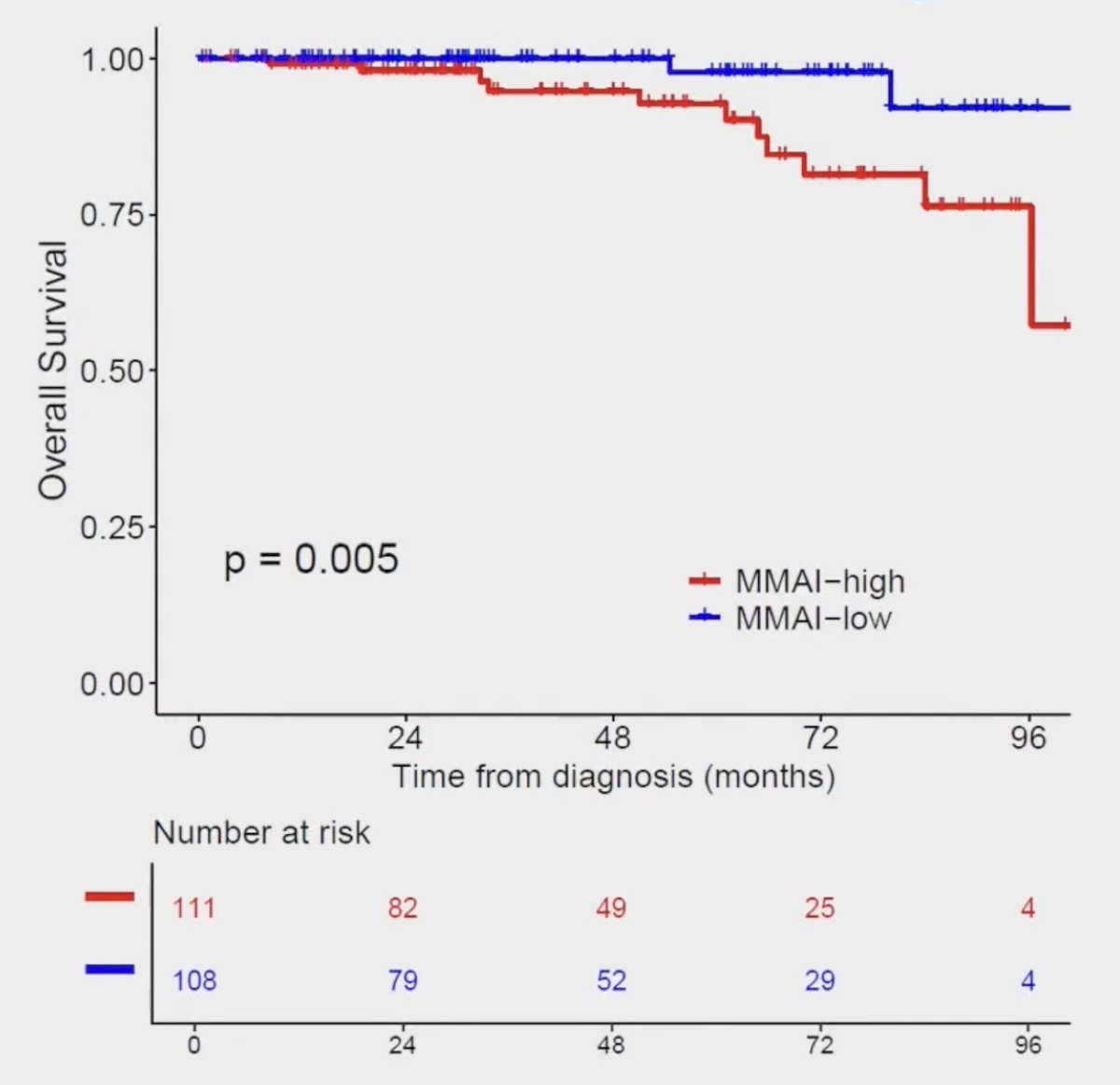
In the STOMP and ORIOLE subset (n = 51; median follow-up of 61 months), MMAI was not prognostic for metastasis-free survival (HR 1.24, 95% CI 0.64-2.43, p = 0.52). MMAI however was predictive for metastasis-directed therapy benefit as patients with high (HR 0.32, 95% CI 0.12-0.90; p = 0.03), but not low (HR 1.59, 95% CI 0.63-4.04; p = 0.33) MMAI demonstrated improvement in metastasis-free survival when treated with metastasis-directed therapy (p-interaction = 0.02):
Dr. Sutera concluded his presentation discussing validation of a digital pathology-based multimodal artificial intelligence model in oligometastatic castration-sensitive prostate cancer with the following take-home points:
- This study has shown for the first time that the ArteraAI MMAI biomarker is prognostic for overall survival in patients with oligometastatic castration-sensitive prostate cancer
- Further, MMAI appears to predict benefit of metastasis directed therapy with high MMAI demonstrating a greater improvement in metastasis-free survival following metastasis directed therapy over observation.
- Further work in validating these findings is warranted to allow for greater personalization in the management of patients with oligometastatic castration-sensitive prostate cancer.
Presented by: Philip Sutera, MD, Radiation Oncologist, Johns Hopkins Radiation Oncology Kimmel Cancer Center, Baltimore, MD
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Associate Professor of Urology, Georgia Cancer Center, Wellstar MCG Health, @zklaassen_md on Twitter during the 2024 American Society for Radiation Oncology (ASTRO) Annual Meeting, Washington, DC, Sun, Sept 29 – Wed, Oct 2, 2024.
Related content: MMAI Score Prognostic for Overall Survival in Oligometastatic Castration-Sensitive Prostate Cancer - Phuoc Tran & Tim Showalter
References:
- Ost P, Reynders D, Decaestecker K, et al. Surveillance of metastasis-directed therapy for oligometastatic cancer recurrence: A prospective, randomized, multicenter phase II trial. J Clin Oncol. 2018 Feb 10;36(5):446-453.
- Phillips R, Shi WY, Deek M, et al. Outcomes of Observation vs Stereotactic Ablative Radiation for Oligometastatic Prostate Cancer: The ORIOLE Phase 2 Randomized Clinical Trial. JAMA Oncol 2020 Mar 26;6(5):650-659.
- Esteva A, Feng J, van der Wal D, et al. Prostate cancer therapy personalization via multi-modal deep learning on randomized phase III clinical trials. NPJ Digit Med. 2022 Jun 8;5(1):71.
- Spratt DE, Tang S, Sun Y, et al. Artificial Intelligence Predictive Model for Hormone Therapy Use in Prostate Cancer. NEJM Evid 2023;2(8).
- Spratt DE, Liu VYT, Jia AY, et al. Meta-analysis of Individual Patient-level Data for a Multimodal Artificial Intelligence Biomarker in High-risk Prostate Cancer: Results from Six NRG/RTOG Phase 3 Randomized Trials. Eur Urol. 2024 Oct;86(4):369-371.


