(UroToday.com) The 2024 American Urological Association (AUA) annual meeting held in San Antonio, TX between May 3 and May 6, 2024, was host to the Bladder Cancer Invasive Podium Session. Dr. Hayato Yamamoto presented the results of a retrospective multicenter study exploring the association between cutaneous reactions related to treatment with Enfortumab Vedotin and its relationship with serum albumin levels in patients with advanced urothelial carcinoma.
Dr. Yamamoto opened his talk by showing that Enfortumab vedotin (EV), which is a novel antibody-drug conjugate directed against nectin-4 is approved for the treatment of advanced urothelial cancer refractory to prior therapies,1 and most recently for untreated advanced urothelial carcinoma.2 He went on to show the treatment algorithm for advanced urothelial carcinoma in Japan, placing EV after progression of first-line chemo, and either maintenance avelumab or pembrolizumab (Algorithm below).
The aim of their study was to explore the occurrence of cutaneous reactions and their potential risk factors in patients receiving EV treatment for advanced urothelial cancer. This study uses a retrospective multicentre dataset. Data were analyzed for patients treated between December 2021 and June 2023. For this analysis, they included 56 patients treated across multiple institutions and with a median follow-up of 11 months. The median age of the cohort was 73 years and most of them (73%) were male. Patient characteristics are shown in the table below:
In terms of oncological outcomes, 37% of the patients experienced treatment response and the median overall survival was 9 months.
Dr. Yamamoto went on to show that 89% of patients treated with EV experienced adverse events. However, only 27% of cases reported Grade 3 or higher events. Skin rash was the most frequent adverse effect, observed in 57% of cases, with Grade 3 or higher skin reactions occurring in 11%.
They conducted univariate analyses, revealing that the following factors were significant risk factors for developing skin reactions:
- Female sex (p=0.037)
- Good performance status (p=0.038)
- Shorter height (p=0.012)
- Higher serum albumin levels (p=0.008)
- Lower C-reactive protein levels (p=0.0017)
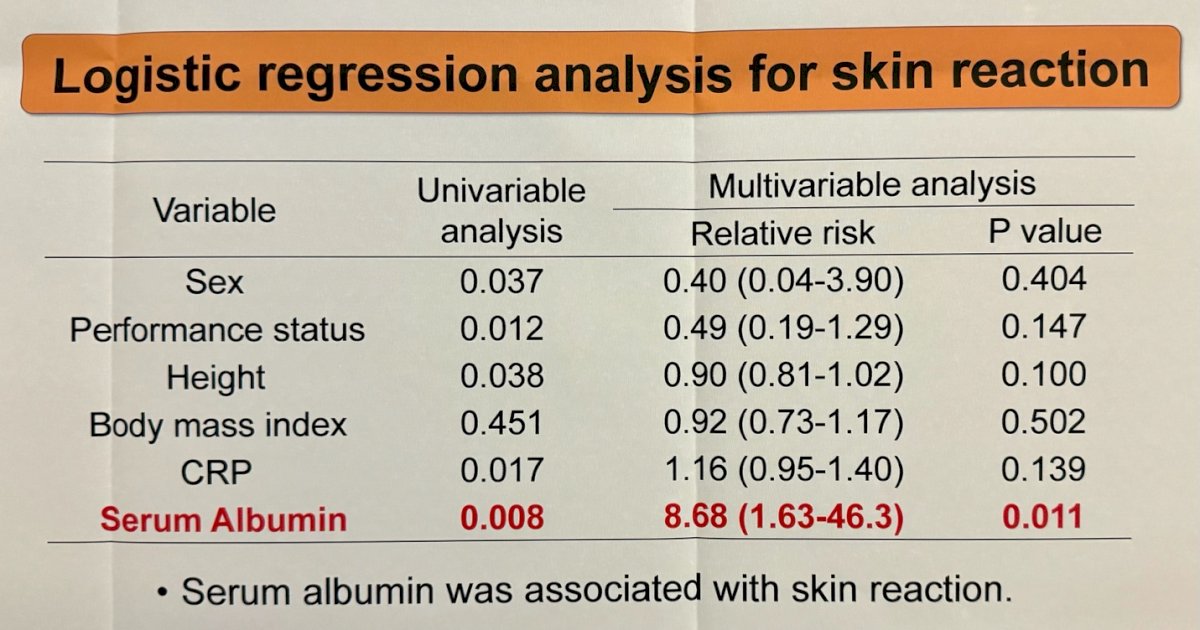
Patients with cutaneous reactions had median serum albumin values of 3.6 mg/dL, compared to 3.0 mg/dL in patients without skin reactions. However, in the multivariate logistic regression analysis, high serum albumin levels were strongly associated with the development of skin reactions (HR=8.68, 95% CI=1.64 – 46.3), and no other factor was associated with skin reactions.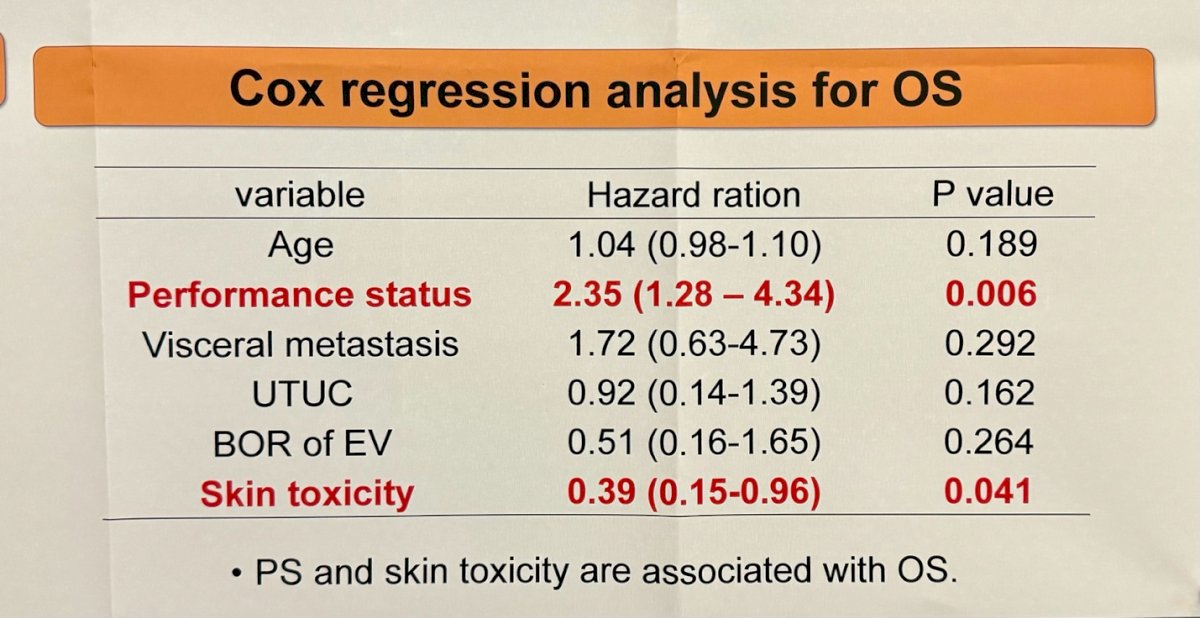
Interestingly, patients experiencing skin reactions exhibited longer overall survival compared to those without reactions (p=0.015), but skin reaction was not associated with progression-free survival.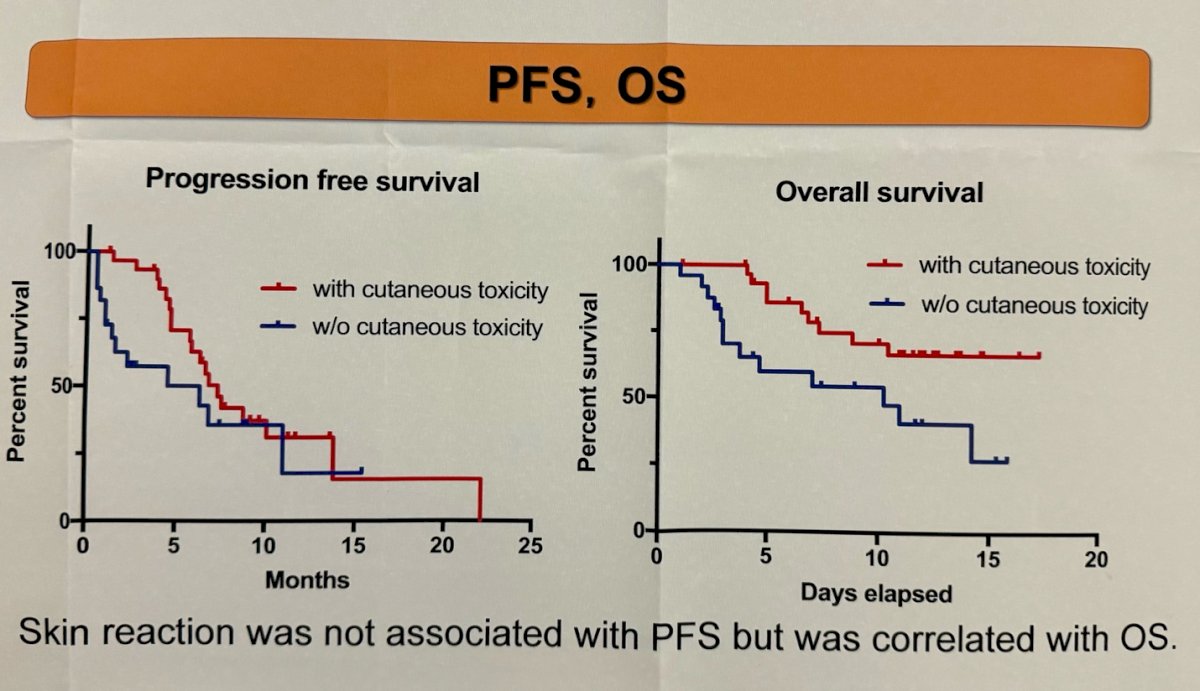
Dr. Yamamoto concluded his presentation by summarizing the following key points:
- Enfortumab vedotin was significantly associated with skin reactions, and skin rash was the most frequent (57% of cases)
- Higher albumin was strongly associated with the occurrence of skin reactions, as indicated by both univariate and multivariate analyses.
- Patients experiencing skin reactions exhibited longer overall survival.
Presented by: Hayato Yamamoto MD, Department of Urology, Hirosaki University School of Medicine, Hirosaki, Japan
Written by: Julian Chavarriaga, MD - Society of Urologic Oncology (SUO) Clinical Fellow at The University of Toronto, @chavarriagaj on Twitter during the 2024 American Urological Association (AUA) annual meeting held in San Antonio, TX between May 3rd and May 6th, 2024
References:
- Powles, Thomas, et al. Enfortumab vedotin in previously treated advanced urothelial carcinoma. New England Journal of Medicine 384.12 (2021): 1125-1135.
- Powles Thomas, et al. Enfortumab Vedotin and Pembrolizumab in Untreated Advanced Urothelial Cancer. N Engl J Med. 2024 Mar 7;390(10):875-888.


