Barcelona, Spain (UroToday.com) Androgen deprivation therapy (ADT) has long been considered the standard of care for patients with metastatic prostate cancer, however recent studies have shown improved survival outcomes by adding additional agents. In the TITAN study, compared with placebo, the addition of apalutamide to ADT significantly improved radiographic progression-free survival (rPFS; HR 0.48, 95% CI 0.39-0.60; p < 0.0001), and overall survival (OS; HR 0.67, 95% CI 0.51-0.89; p = 0.0053) in patients with mCSPC.1 Dr. Agarwal and colleagues evaluated pain, fatigue, and overall health-related quality of life of patients in TITAN.
For TITAN, 1,052 men were randomized 1:1 to receive apalutamide (240 mg/d) or placebo, added to ADT, in 28-day cycles. Patients were stratified by Gleason score at diagnosis (≤ 7 vs > 7), region (North America vs European Union vs other countries), and prior docetaxel use (yes vs no). Patient reported outcomes were assessed using the Brief Pain Inventory-Short Form (BPI-SF), Brief Fatigue Inventory (BFI), Functional Assessment of Cancer Therapy-Prostate (FACT-P), and Euro QoL Group EQ-5D-5L. BPI and BFI were completed for 7 consecutive days (day-6 plus day 1 of each cycle visit), then at months 4, 8, and 12 in follow-up. FACT-P and EQ-5D-5L were completed cycle 1 through cycle 7, then every other cycle through end of treatment, and at months 4, 8, and 12 in follow-up. Analyses included descriptive statistics and mean change from baseline using mixed model of repeated measures.
Not surprisingly, patients were relatively asymptomatic at baseline: on 0-10 severity scales, median pain scores were 1.14 for apalutamide and 1.00 for placebo, and median fatigue scores were 1.29 for apalutamide and 1.43 for placebo. Patient experience of pain and fatigue was similar between groups for the duration of treatment. For the majority of patients in both arms, pain and fatigue remained stable or improved during treatment, with greater improvements observed in patients with higher baseline severity scores: 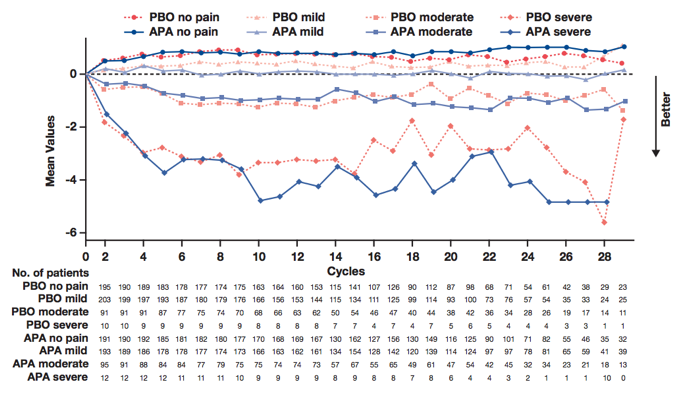
Although not statistically significant, time to pain progression favored apalutamide over placebo:
FACT-P total score and EQ-5D-5L data showed similar maintenance of overall health-related quality of life in both arms. Similar tolerability was experienced between groups based on the FACT-P single item side effects bother question:
Dr. Agarwal concluded his presentation of the TITAN patient reported outcomes with the following conclusions:
- In addition to the substantial benefits in the dual TITAN primary end points of OS and rPFS demonstrated previously, overall health-related quality of life was preserved with the addition of apalutamide to ADT in patients with mCSPC
- These data support the addition of apalutamide to ADT for a broad range of patients with mCSPC
Clinical trial identification NCT02489318
Presented by: Neeraj Agarwal, MD, Associate Professor in the Division of Oncology, Department of Medicine, University of Utah School of Medicine. Director of the Genitourinary Oncology Program in the Oncology Division, the Co-leader of the Urologic Oncology Multidisciplinary Program and the Associate Director of Clinical Trials at the Huntsman Cancer Institute.
Co-Authors: N. Agarwal 1, K. Mcquarrie 2, A. Bjartell 3, S. Chowdhury 4, A. Gomes 5, B.H. Chung 6, M. Özgüroğlu 7, Á. Juárez Soto 8, A. Merseburger 9, H. Uemura 10, D. Ye 11, R. Given 12, B. Miladinovic 13, L. Dearden 14, K. Deprince 15, V. Naini 16, A. Lopez-Gitlitz 17, K. Chi 18
2. Janssen Research & Development, Horsham, US
3. Skåne University Hospital Malmö, Malmö, SE
4. Guy's and St. Thomas' Hospital NHS Trust, London, UK
5. Liga Norteriograndense contra o cancer, Natal, BR,
6. Yonsei University College of Medicine and Gangnam Severance Hospital, Seoul, KR
7. Istanbul University - School of Medicine, Istandul, TR
8. Hospital Universitario de Jerez de la Frontera, Cadiz, ES
9. University SH.-Lübeck, Lübeck, DE
10. Kindai University Faculty of Medicine, Osaka, JP
11. Fudan University - Shanghai Cancer Center (FUSCC), Shanghai, CN
12. Urology of Virginia, Eastern Virginia Medical School, Norfolk, US
13. Janssen Research & Development, San Diego, US
14. Janssen, High Wycombe, UK
15. Janssen EMEA Belgium, Beerse, BE
16. Janssen Research and Development, San Diego, US
17. Janssen Research & Development, Los Angeles, US
18. BC Cancer Agency - Vancouver, Vancouver, CA
Written by: Zachary Klaassen, MD, MSc – Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia Twitter: @zklaassen_md at the 2019 European Society for Medical Oncology annual meeting, ESMO 2019 #ESMO19, 27 Sept - 1 Oct 2019 in Barcelona, Spain
Reference:
Further Related Content:
Health-Related Quality Of Life After Apalutamide Treatment In Patients with Metastatic Castration-Sensitive Prostate Cancer (TITAN): A Randomised, Placebo-Controlled, Phase 3 Study


