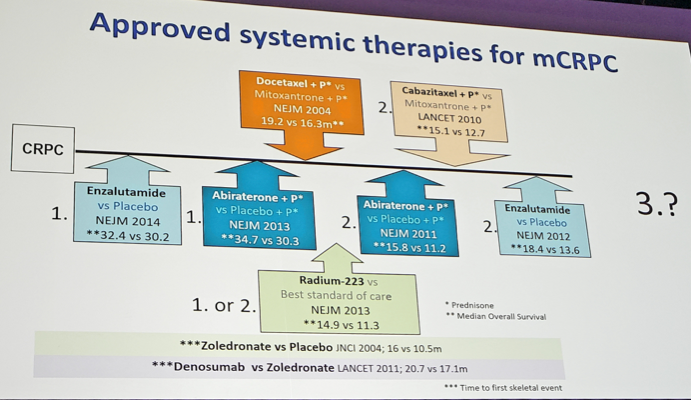
Of note, a clear-cut 3rd line agent has not been approved. Despite several agents being approved for mCRPC, the optimal sequence as to when to give these medications is unclear. None of the mCRPC trials have compared the new agent with current standard therapies. The best treatment option after docetaxel and one of the novel endocrine agents is unknown, representing an unmet clinical need. Should we switch the androgen receptor therapies? Should we use a cytotoxic chemotherapy? This is the backdrop for a third line clinical trial.
Dr. Gillessen notes that the two arms of the trial were well balanced and had a high proportion of men ≥75 years of age: 34.9% in the cabazitaxel arm and 27.0% in the adjuvant radiotherapy (ART) arm. Additionally, 66.7% of patients in the cabazitaxel arm had pain, compared to 71.4% of patients in the ART arm. Thus, this is a representative population of patients with heavily pretreated mCRPC, focusing on patients with a “shortish” (≤12 month) response to first ART.
The efficacy data is impressive, with improved progression-free survival (PFS) favoring cabazitaxel at a median PFS of 8.0 months vs 3.7 months in the ART arm (hazard ratio (HR) 0.54, 95% confidence interval (CI) 0.40-0.73). This finding was consistent across key subgroups, especially timing of ART with respect to receipt of docetaxel, as well as time from ART initiation to progression. However, based on what was reported today, we do not know what the impact of docetaxel in the castration-sensitive setting vs the castration-resistant setting is. Additionally, cabazitaxel significantly improved OS at a median 13.6 months vs 11.0 months for ART: HR 0.64, 95% CI 0.46-0.89. In the context of other smaller studies in this disease space, the results of CARD generally compare favorably:
With regards to cabazitaxel dose, there is room for dose reduction from the dose given in CARD (25 mg/m2 IV Q3W + prednisone + G-CSF). This is taken from the PROSELICA study, which showed that 20 g/m2 IV Q3W showed no difference in OS compared to the traditional dosing of 25 mg/m2 IV Q3W1.
Dr. Gillessen concluded her discussion of CARD with several key messages: (i) It was a well-designed trial addressing an unmet clinical need; (ii) It included a patient population representative of patients seen in the clinic after docetaxel and one line of androgen receptor therapy; (iii) The current treatment landscape should be for fit patients to receive docetaxel and abiraterone or enzalutamide at some stage (+/- radium-223). A subgroup of patients can derive a benefit from poly ADP-ribose polymerase (PARP) inhibitors and another subgroup can derive benefit from pembrolizumab; (iv) Based on information presented in the CARD trial, cabazitaxel is a new standard of care for 3rd line patients with progressive disease on prior androgen receptor therapy <=12 months.
Several unanswered questions include:
- Can we extrapolate to patients receiving agents in the castration-sensitive stage?
- What about patients with performance status of 2 or worse?
- What about patients responding to prior androgen receptor therapy >12 months?
Reference:
- Eisenberger, Mario, Anne-Claire Hardy-Bessard, Choung Soo Kim, Lajos Géczi, Daniel Ford, Loïc Mourey, Joan Carles et al. "Phase III study comparing a reduced dose of cabazitaxel (20 mg/m2) and the currently approved dose (25 mg/m2) in postdocetaxel patients with metastatic castration-resistant prostate cancer—PROSELICA." Journal of Clinical Oncology 35, no. 28 (2017): 3198-3206.
Cabazitaxel versus Abiraterone or Enzalutamide in Metastatic Prostate Cancer - Beyond the Abstract


